Your shopping cart is empty!
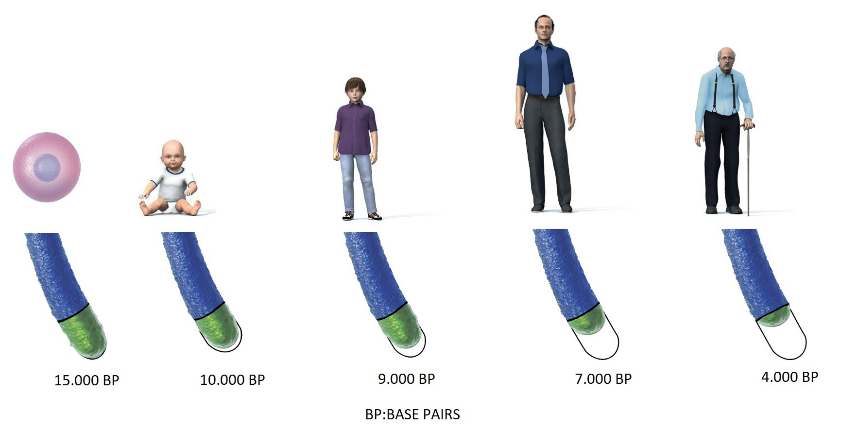
Telomeres work like a timekeeper of each cell. Starting from a zigot cell, generally telomere length is approximately 15.000 Base Pairs (BP). There are huge number of cell replications in the womb so when a baby is born telomeres are shortened to 10.000 BP. Even in a womb cellular aging begins. We can consider this telomere length as a life and health credit. Each individual has different credit coming from mother, father ancestors.
This as a credit from the moment we are born and every one of us has different way to spend our telomere credit (very similar to how we spend our money). There are good and bad habits that effects the telomere shortening (will be explained in a different topic but very briefly smoking is a very bad habit for your telomeres while exercise is beneficial). Eventually each person is aged totally different speed than the others. Even for the single cell twins has different speed of aging and health concerns after certain age.
Life genarally starts with 10.000 BP and ends with 4.000 BP due to old age. The difference gives us 6.000 BP credit for whole our life in between. On the other hand when a cell is replicating itself, it consumes 50 BP long telomeres under normal conditions. During replication process DNA copies itself perfectly while protective end tips of DNA can not be copied completely. Therefore each new copy of a cell is older than previous one because of telomere shortening.
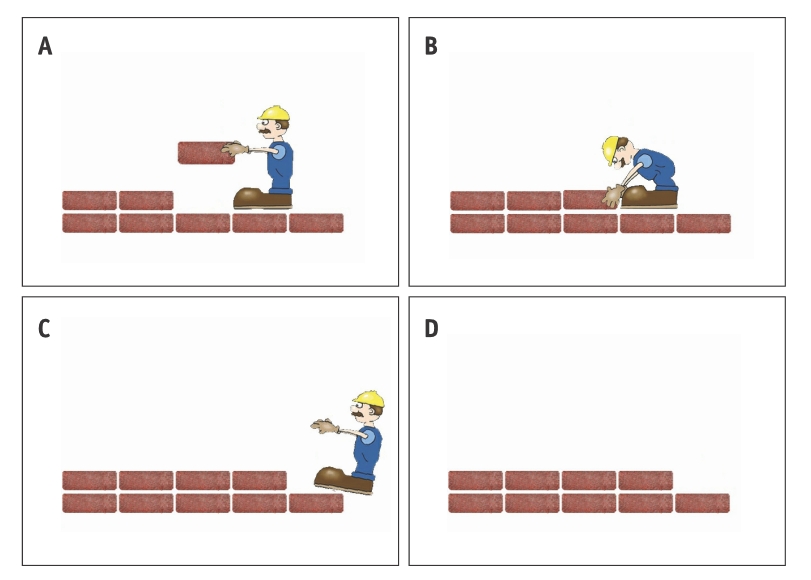
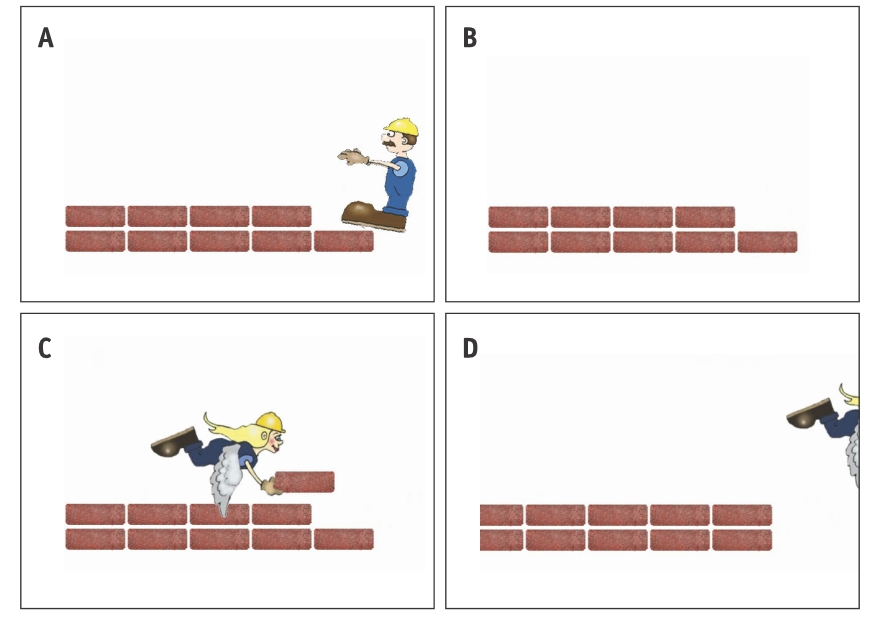
As an analogy, think of a DNA as a long row of bricks, and of DNA replication as a bricklayer walking backwards on top of a brick wall laying a new layer on top of that row. When the end of the wall is reached, the bricklayer finds himself standing on top of the brick he’s supposed to replicate. Since he can’t put down a new brick where his feet are, he steps back and falls off the wall – leaving the very end of the wall bare. As a result, the new copy of the wall is shorter and older than the previous cell.
If we divide our life credit 6.000 BP to 50BP we find that therotically a human lifespan is about 120 years. And this is practically true as well. According to records longest living human was 122 year of French woman named Jeanne Calment.
This phenmenon is known as Hayflick limit. This was discovered by Dr. Leonard Hayflick in 1961 but he could not explained the reason at this time. But after his works, in 2009 this was explained by three scientists Elisabeth Blackburn, Carol Greider and Jack Stozak and they won the Nobel Prize by explaining telomere and telomerase relation.
The body has its own mechanism to protect telomere length: the enzyme telomerase. Telomerase is an enzyme that adds nucleotides to telomeres, extending these DNA end caps, and hereby extending the Hayflick Limit.
In the 1980s scientists studying telomeres (caps at the end of each strand of DNA) discovered telomerase (the enzyme that maintains telomere length). The research of these scientists revolutionized our understanding of cellular aging-and its responsibility for cell and organ malfunction.
This pioneering research was so important that the Nobel Prize in Physiology/Medicine was awarded in October 2009 for the breakthrough discovery of telomerase.
In early genetic research, nucleotide molecules found at the ends of the chromosomes were regarded as non-functioning “garbage DNA.” We now know that’s hardly the case. Shortening of these DNA sequences, called telomeres, acts as an aging clock and explains the Hayflick Limit – the number of times cells can divide before becoming non-functioning (senescent) or dying (apoptosis).
To understand what telomerase is doing, we need to know how some cells in our body keep theirself young and almost immortal.
In our bodies there are 3 main group of cells:
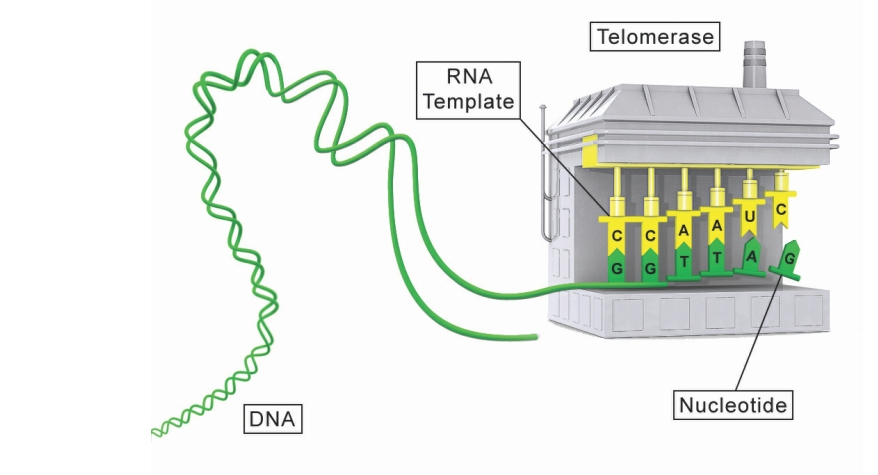
First two group of cells simply do not age just because they have abundant telomerase due to abilty to produce telomerase. In germ (reproductive) cells, a protein-coding gene that activates telomerase, Telomerase Reverse Transcriptase (TERT), is permanently switched on. Stem cells and some rapidly dividing cells also have telomerase, but, unlike in germ cells, the gene is not turned on permanently and the amount of telomerase declines with age. Even the high number of replication of sperm (except women germ cells) and stem cells are able to keep their telomeres long because they are able to produce telomerase enyzme when the telomeres get shorter after replication. This is managed by a gene in DNA. And we know that DNA information is same in each cell.
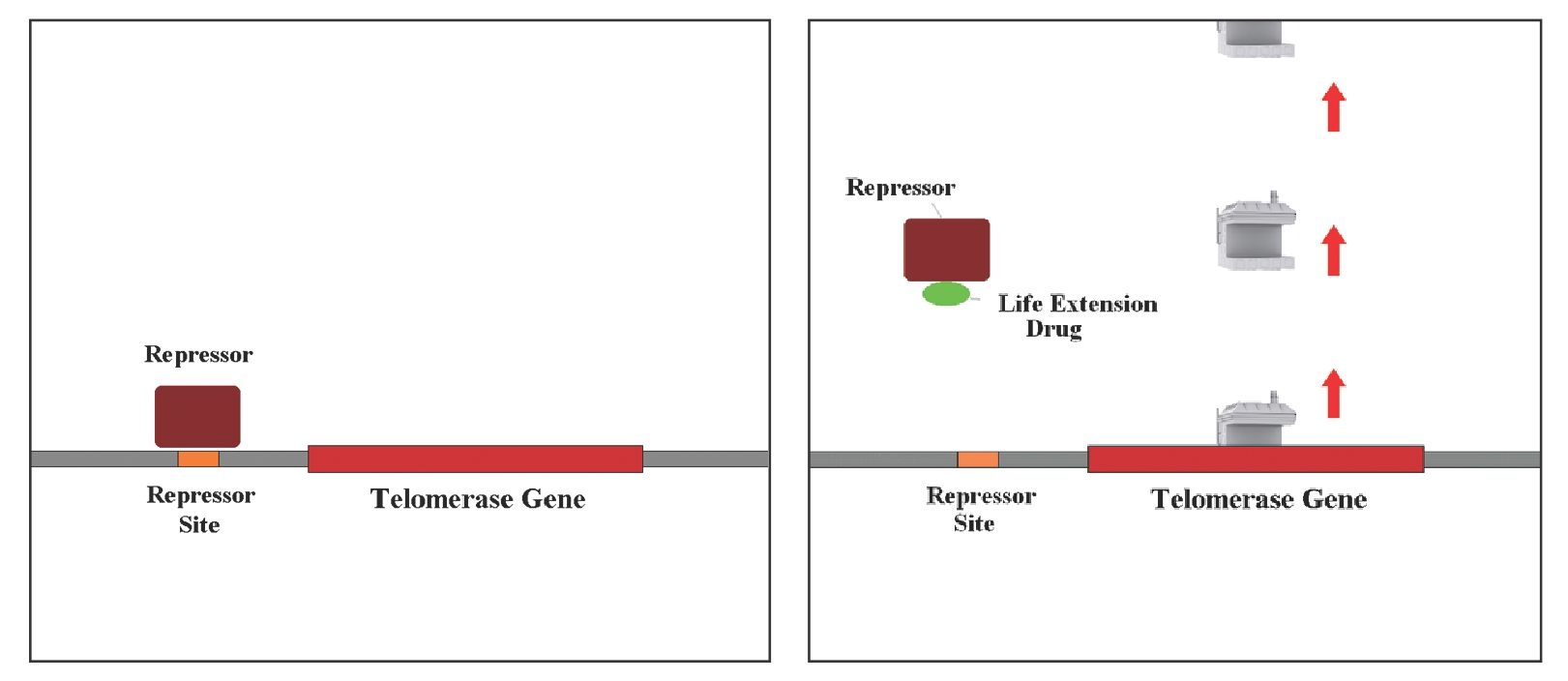
Then what makes somatic cells telomeres shorten? Unfortunately somatic cells do not have such ability only because most of our cells don’t express telomerase is that the gene responsible for this purpose is repressed. The problem is that the vast majority of somatic (body) cells have the TERT gene turned off. As cells lose their ability to make enough telomerase, they cannot replenish worn out cells and that eventually leads to compromised organ function.
There are one or more regions of DNA neighboring the telomerase gene that serve as binding sites for a protein, and, if that protein is bound to them, telomerase will not be stimulated and telomeres get shorter by each replication. This mechanism makes the cells age and non-functional.
When a cell has a critcally short telomere cell has to take an action. In a cell there are 92 telomeres and cell takes a decision according to the shortest telomere among 92 in a cell. Cell may have 91 long telomeres and just single very short telomere. Then the decision will be taken based on this shortest telomere. Therefore in a cell the shortest telomeres have the highest impotance.
A cell has to decide 3 main options;
In this case keeping the telomeres as long as possible is very critical for cell functions.
Short answer is yes. But there are several points to explain about telomere lenghtening as well as keeping the telomeres long.
Once we define the problem we may discuss the solution possibilities. If the problem was telomere shortening due to lack of telomerase, then we can develop some approaches to keep the telomeres long as well as lenghtening them.
For an adult body we have almost 100 trillion cells and luckly all DNA coding is same in these cells. This gives us a huge advantage because if some cells are able to stimulate the telomerase then the other should be able to. There are several actions to be taken to make the cells young and functional again.
Telomerase activator food supplement TA-65 that removes the repressing protein in somatic cell DNA’s. Therefore activates telomerase transiently.
On the other hand scientists are working on a gene theraphy to activate telomerase in all our cells to rejuvenate whole body. And there are very promising studies while this method is very immature yet very expensive now (> 3 million USD/treatment).